Learn about the nursing implications of these innovative devices.
Takeaways:
- The leadless pacemaker is a right ventricular single chamber pacemaker used to treat symptomatic bradycardia.
- The leadless pacemaker eliminates the complications of conventional pacemakers related to leads and surgical pocket complications.
- Currently, leadless pacemakers can pace only from the right ventricle.
ONE of the most recent developments in cardiac pacing is the leadless pacemaker (LP). This device is one of the biggest advancements in pacemakers since they came into use for symptomatic bradycardia more than 50 years ago. (See Traditional pacemaker basics.) However, as with any new technology, a knowledge gap exists. Many nurses may not have encountered a patient with an LP and little nursing literature on the topic exists. Understanding this new technology will help you deliver safe care to patients who may be candidates for an LP or who may already have one.
LPs vs. traditional pacemakers
The LP was developed to avoid the complications associated with the leads and surgical pocket required when placing a traditional pacemaker. LPs, which are miniature devices, are implanted directly into the right ventricle via the femoral vein in the groin.
LPs are single-chamber devices, so currently they’re available only for a limited number of pacemaker candidates. The 2009 world survey by Mond and Proclemer provides prevalence data on the use of pacemakers by type. In the United States, just 20% of patients who required permanent pacemakers received traditional single-chamber devices, while the remaining 80% received traditional dual-chamber devices. (See Pacemaker indications.)

LP availability
Medtronic won Food and Drug Administration (FDA) approval to market the Micra LP on April 6, 2016. (See Pacemaker comparison.) The device is 25.9 mm long, 6.7 mm wide, and weighs 2 grams. It’s inserted with a catheter through the femoral vein with a 23 French introducer, advanced into the right ventricle, then attached to the endocardium. After it’s fixed to the inside of the right ventricle, the catheter and introducer are removed. Vascular closure of the femoral vein, using manual pressure, purse-string suture, or a vascular closure device, prevents bleeding at the groin site. As of October 2017, Medtronic reports that more than 10,000 patients in 40 countries have a Micra LP.
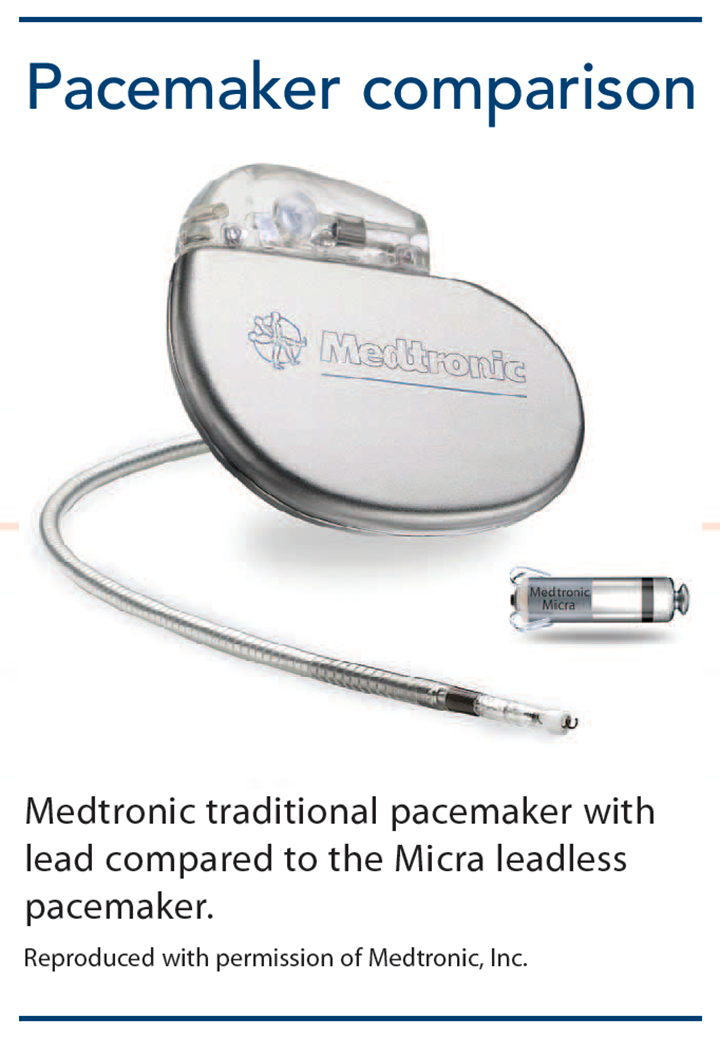
Currently, Micra LP is the only LP on the market. St. Jude Medical (now Abbott) conducted clinical trials of its Nanostim LP, but after 1,423 implants, the company halted implantation in October 2016 because of seven reports of battery issues that resulted in lost telemetry and pacing output. The manufacturer recommends device replacement for anyone with the Nanostim LP starting with those who’ve had the pacemaker for the longest duration. To date, 34 patients have experienced battery depletion with the Nanostim. Of those, six experienced bradycardia. No other adverse outcomes have been reported. Crotti reports that Abbott is now planning to focus on developing the dual-chamber leadless pacemaker, as this is where the greater need is. Micra’s battery life is expected to be 10 years or more.
LP candidates
Patients who require a VVIR (ventricle pacing, ventricle sensing, paced beat inhibition, rate modulation) for symptomatic bradycardia may be candidates for the LP. (Note: For pacing code information, visit myamericannurse.com/?p=52458.) Patients with existing pacemakers who’ve had surgical pocket or trans – venous lead complications also may benefit from this technology.
Some patients aren’t good candidates for an LP. They may require the more complex features and programmable technologies available in dual-chamber and biventricular pacemakers. Others may have a mechanical tricuspid valve, which prevents passing the LP into the right ventricle. Similarly, if a patient has a vena cava filter, the introducer can’t be inserted through the groin. (See LP contraindications.)

Nursing care
Most LP placement procedures take about 1 hour, and patients usually stay in the hospital overnight.
After device placement, assess the patient’s blood pressure, heart rate, cardiac rhythm, respiratory rate, and oxygen saturation; evaluate the groin site for bleeding, oozing, or hematoma; and assess circulation by palpation or Doppler sig nals distal to the procedure site. Monitor the patient every 15 minutes for 1 hour, every 30 minutes for 1 hour, every hour for 2 hours, and then follow the usual protocol unless assessments indicate more frequency is needed. Bedrest may be maintained for about 4 hours to allow for hemo – stasis at the groin site and to prevent bleeding complications.
The appropriate provider will interrogate the pacemaker by placing a wand-like device over the pacemaker area to allow the pacemaker to communicate with a computer that evaluates device function, the battery, and any abnormal cardiac rhythms the patient may have experienced.
Notify the provider of any assessments that are out of normal range or unexpected, such as vital sign changes, chest pain or palpitations, abnormalities on the cardiac monitor, diminished pulses, and color change or temperature change of the extremity with the groin site. A failure to pace will appear on the cardiac monitor as an absent pacemaker spike when one is expected and as a heart rate lower than the rate set on the pacemaker. Note that if the patient’s native heart rate is higher than the demand rate on the pacemaker, then no pacing will occur and no pacemaker spikes will be seen on the monitor.
If you observe a failure to pace when pacing should occur, assess the patient clinically for symptoms of bradycardia and notify the provider or rapid response team. Interventions will depend on the patient’s clinical status and may include emergency measures for symptomatic bradycardia, such as atropine, dopamine, or epinephrine; a temporary pacemaker; continued observation; and bed rest. Other pacemaker rhythm disturbances include a failure to capture (the pacemaker fires, but doesn’t cause contraction), oversensing (pacemaker fires less frequently than it should), and undersensing (pacemaker fires more frequently than it should).
Alert the provider if you find evidence of hematoma, bleeding, or pain at the affected groin site. While awaiting evaluation from the provider, demarcate the area on the dressing, reinforce the dressing as necessary, and apply pressure if needed.
Potential complications
At the start of the investigational study for both the Micra LP and Nanostim LP, the FDA identified potential complications, including right ventricle perforation, cardiac tamponade, pneumothorax, hemothorax, device dislodgement, device migration during implantation, infections requiring reoperation or extraction, and femoral vascular access complications (arteriovenous fistula, pseudo – aneurysm, and excessive bleeding).
 At the completion of the investigational trial period, Reynolds and colleagues reported that the Micra was successfully implanted in 719 of 725 patients. Twenty-eight major complications occurred in 25 of the 725 patients (a rate of 4%). These complications included one with pulmonary embolism, one with deep vein thrombosis, five with events at the groin puncture site, 11 with cardiac injury (three with perforation, eight with pericardial effusion), and eight with other cardiac issues (one with myocardial infarction, three with cardiac failure, one with pacemaker syndrome, and one each with metabolic acidosis, presyncope, and syncope).
At the completion of the investigational trial period, Reynolds and colleagues reported that the Micra was successfully implanted in 719 of 725 patients. Twenty-eight major complications occurred in 25 of the 725 patients (a rate of 4%). These complications included one with pulmonary embolism, one with deep vein thrombosis, five with events at the groin puncture site, 11 with cardiac injury (three with perforation, eight with pericardial effusion), and eight with other cardiac issues (one with myocardial infarction, three with cardiac failure, one with pacemaker syndrome, and one each with metabolic acidosis, presyncope, and syncope).
Since the Micra has been approved by the FDA, Roberts and colleagues have been reporting on the progress of the LP in the real world and comparing their results with those of the investigational research. In this ongoing study, LPs have been implanted in 792 of 795 patients. Thirteen major complications occurred in 12 patients (a rate of 1.51%). These complications included one deep vein thrombosis, six events at the fem oral access site, one cardiac perforation, one device dislodgment, one pacing issue, and one each with chest pain, pulmonary edema, and sepsis.
Although complication rates are low and therefore encouraging, nurses must be vigilant and ready to act when they occur. Be alert for symptoms of right ventricular perforation (chest pain, dyspnea, abdominal pain, hiccoughs, hemodynamic instability, cardiac tamponade, pacing failures, loss of consciousness, heart failure, and cardiac arrest). Anticipate that a chest x-ray, echo cardiogram, and chest computed tomography scan may be ordered for any patient with suspected right ventricle perforation.
Symptoms of cardiac tamponade are anxiety, restlessness, dyspnea, tachycardia, tachypnea, cool skin, diaphoresis, and Beck’s triad (muffled heart sounds, increased jugular venous distention, and hypotension). If cardiac tamponade is suspected, anticipate the need for an echocardiogram and a pericardiocentesis. These medical emergencies require a call for the cardiac team stat or the rapid response team. Anticipate that the patient will go to the intensive care unit and may need emergency surgery.
Discharge teaching
Before the patient is discharged from the hospital, the provider will evaluate the pacemaker for proper pacing, a chest x-ray will be ordered to verify device position, and a 12-lead electrocardiogram (ECG) will be performed to verify cardiac rhythm. Inform the patient that if he or she is feeling well, a return to usual activities can begin in 1 or 2 days. To allow for groin healing, the patient should avoid heavy lifting and squatting for 1 week.
Discuss the importance of attending follow-up appointments for a physical exam, ECG, and pacemaker adjustments as needed to pace the heart faster or slower or to deliver more or less energy to optimize therapy. Explain that the pacemaker should relieve the symptoms of bradycardia and that the patient should be able to complete activities of daily living more easily.
Instruct the patient to report symptoms of syncope, dizziness, confusion, fatigue, and exercise intolerance. These symptoms may indicate pacemaker malfunction or the need for setting adjustments. Tell the patient to always carry his or her identification (ID) card, which includes important information about the type of pacemaker, serial number, and date and location of implantation.
Verify that the patient has the device patient manual, which includes information about the effects of electromagnetic interference (EMI). Although cell phones are described as having low or no risk, instruct the patient not to carry his or her cell phone in a shirt pocket. Explain to the patient that if he or she feels dizzy or experiences a rapid or irregular heartbeat while using an electrical device, to let go of the device and move away from it. The pacemaker should return to normal function away from the EMI. In addition, explain that the patient shouldn’t enter an area with a “no pacer” symbol.
Patients with an LP should talk with their cardiologist before undergoing medical procedures. Electrosurgery, electrocautery, external defibrillation, lithotripsy, or radiation therapy can cause interference with pacemaker functioning.
Medtronic describes its LP as being magnetic resonance imaging (MRI) conditional. “Conditional” refers to the MRI magnet strength, proper pacemaker functioning, duration of implant, and country in which the pacemaker was inserted. These conditions are subject to change and will evolve over time with this new device. The Medtronic MRI technician manual defines the conditions.
Although airport security metal detectors aren’t likely to affect an LP, Medtronic recommends that patients pass through the detectors at a normal pace but not linger in the archway. Advise the patient to immediately move out of range of the detector if he or she experiences fatigue or dizziness. If the metal detector is set off by the LP, the patient should show the ID card to security personnel and explain that the hand – held detection wand should not be waved over the device. Patients also can show their ID card and request alternative screening.
Keeping pace with technology
The LP represents a profound change in pacemaker technology. More literature is needed to provide nurses with the education they need to keep up with these innovations. Currently, LPs are limited to right ventricular pacing; however, other implanted cardiac devices are being investigated that will work in combination with the LP for use in other areas of the heart.
Joyce Bulger-Noto is a nursing professional development specialist at New York–Presbyterian Weill Cornell Hospital in New York.
Selected references
Bernstein AD, Daubert JC, Fletcher RD, et al. The revised NASPE/BPEG generic code for antibradycardia, adaptive-rate, and multisite pacing. Pacing Clin Electrophysiol. 2002;25(2):260-4.
Crotti N. Abbott is developing a dual-chamber leadless pacemaker. June 4, 2018. medicaldesignandoutsourcing. com/abbott-is-developing-a-dual-chamber-leadless-pacer/
Epstein AE, DiMarco JP, Ellenbogen KA, et al. ACC/AHA/HRS 2008 Guidelines for device- based therapy of cardiac rhythm abnormalities: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (writing committee to revise the ACC/AHA/NASPE 2002 guideline update for implantation of cardiac pacemakers and antiarrhythmia devices): Developed in collaboration with the American Association for Thoracic Surgery and Society of Thoracic Surgeons. Circulation. 2008;117(21):e350-408.
Leier M. Advancements in pacemaker technology: The leadless device. Crit Care Nurse. 2017;37(2):58-65.
Medtronic. Micra MC1VR01 Clinician Manual. manuals. medtronic.com/wcm/groups/ mdtcom_sg/@emanuals/@era/@crdm/documents/ documents/contrib_231758.pdf
Mond HG, Proclemer A. The 11th world survey of cardiac pacing and implantable cardioverter- defibrillators: Calendar year 2009—A World Society of Arrhythmia’s project. Pacing Clin Electrophysiol. 2011;34(8):1013-27.
Reddy VY, Knops RE, Sperzel J, et al. Permanent leadless cardiac pacing: Results of the LEADLESS trial. Circulation. 2014;129(14):1466-71.
Reynolds D, Duray GZ, Omar R, et al. A leadless intracardiac transcatheter pacing system. N Engl J Med. 2016;374(6):533-41.
Roberts PR, Clementy N, Al Samadi F, et al. A leadless pacemaker in the real-world setting: The Micra Transcatheter Pacing System Post-Approval Registry. Heart Rhythm. 2017;14(9):1375-9.
Udo EO, Zuithoff NP, van Hemel NM, et al. Incidence and predictors of short- and long-term complications in pacemaker therapy: The FOLLOWPACE study. Heart Rhythm. 2012;9(5):728-35.
United States Food and Drug Administration. Circulatory System Devices Panel Meeting: Leadless Pacemakers. Division of Cardiovascular Devices Office of Device Evaluation, Food and Drug Administration, February 18, 2016. Executive Summary Memorandum General Issues: Leadless Pacemaker Devices. 2016. fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/MedicalDevices/ MedicalDevicesAdvisoryCommittee/CirculatorySystemDevicesPanel/ UCM486733.pdf
ant10-Pacemaker-927















1 Comment.
Global leadless cardiac pacemakers market is expected to reach from US$ 47.1 Mn in 2017 to US$ 269.8 Mn by 2026 expanding at a CAGR of 21.9% from 2018 to 2026.