Learn the risk factors and tailor prevention to device type.
Takeaways:
- Medical device–related pressure injuries (MDRPIs) are a common but preventable patient care issue with simple interventions.
- Prevention guidelines and evidence-based practices can help reduce MDRPIs.
Editor’s note: The author has provided the attached PDF file of a clinical tips card for preventing medical device related pressure injuries. The card is 2 inches by 7 inches; the spot at the top indicates where a hole could be punched to put a laminated version of the card on a lanyard. It could also be carried in the pocket and posted in the unit.
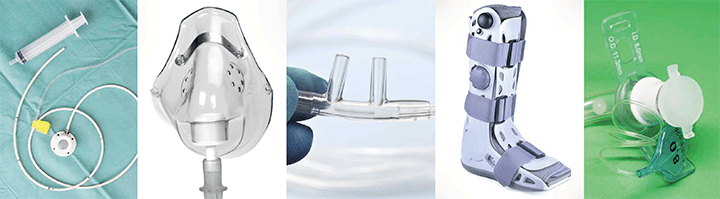 THE Joint Commission’s Quick Safety issue on managing medical device–related pressure injuries (MDRPIs) points out that nearly all hospital patients require at least one medical device for care and treatment, putting them at risk for injury. The National Pressure Ulcer Advisory Panel (NPUAP) defines MDRPI as “localized damage to the skin and/or underlying soft tissue usually over a bony prominence or related to a medical or other device. The injury can present as intact skin or an open ulcer and may be painful. The injury occurs as a result of intense and/or prolonged pressure or pressure in combination with shear.”
THE Joint Commission’s Quick Safety issue on managing medical device–related pressure injuries (MDRPIs) points out that nearly all hospital patients require at least one medical device for care and treatment, putting them at risk for injury. The National Pressure Ulcer Advisory Panel (NPUAP) defines MDRPI as “localized damage to the skin and/or underlying soft tissue usually over a bony prominence or related to a medical or other device. The injury can present as intact skin or an open ulcer and may be painful. The injury occurs as a result of intense and/or prolonged pressure or pressure in combination with shear.”
Nurses can take the lead in preventing MDRPIs by understanding risk factors, tailoring prevention to the device type, collaborating with other team members to ensure continuity of care, and monitoring for change.
Risk
Risk factors associated with MDRPIs include:
- tight device securement that results in poor circulation, friction, or shearing
- poor and/or prolonged patient positioning (pressure for as little as 10 minutes can result in a pressure injury [PI])
- heat that increases perspiration
- moisture under the device that results in macerated skin • edema that increases skin tension
- lack of awareness by staff and patients of preventive measures and PI potential
- failure to routinely assess patients’ skin and devices (skin assessments should be performed at the earliest contact with the patient [in the emergency department or on admission to the unit] and according to the device manufacturer’s recommendations or more often depending on comorbidities and medications).
A 2018 study by Kayser and colleagues found that the most common anatomic locations for MDRPIs are the ears and the feet, and the most common devices associated with MDRPIs include nasal oxygen tubes, casts and splints, and continuous positive airway pressure (CPAP)/bilevel positive airway pressure (BiPAP) masks.
Prevention
Preventing MDRPIs involves reducing pressure or redistributing it over a larger area, controlling the skin’s microclimate, improving patient nutrition, encouraging mobility, and addressing comorbidities. NPUAP prevention guidelines include:
- choosing the correct size medical device to fit the patient
- cushioning and protecting the skin with dressings in high-risk areas (nasal bridge, rim of device)
- removing or moving devices, when possible, to assess skin at least daily
- avoiding device placement over sites of prior or existing PIs
- educating staff about the correct use of devices and skin breakdown prevention
- being cognizant of edema under devices and the potential for skin breakdown (edema can develop after device application, especially in patients receiving massive fluid resuscitation or those with unresolved soft-tissue inflammation)
- confirming that devices aren’t placed directly under a patient who is bedridden or immobile.
Identifying and proactively addressing the cause of an MDRPI can be challenging. (See Early identification.) However, the NPUAP 2014 consensus panel noted that some device-related PIs may be unavoidable when adjusting or moving a device is medically contra – indicated, when a device prevents patient turning or repositioning, and when tissue edema or moisture under a device compromises tissue tolerance and increases the risk for pressure and shear forces.

Prevention measures are unique to the device being used. Consider the following strategies when caring for and monitoring patients.
Nasal cannulas
Nasal cannulas may cause PIs behind the ears, around the nostril, or in the nasal vestibule.
Prevention: Soft nasal cannulas can dramatically lower the incidence of PIs. If a high-flow cannula is needed, protect the back of the ears with a hydrocolloid or foam dressing.
Comment: A quality improvement (QI) project by Lukose and colleagues found that using preventive measures (soft cannulas, hydrocolloid/foam dressing) decreased the incidence of PIs from 5.4% to zero. Lukose’s organization maintained zero PIs related to nasal cannulas for 23 months. In addition to reducing PI incidence, this QI project saved nurses time and has the potential of saving the organization $41,932 per year.
Masks
Masks—including oxygen, CPAP, and BiPAP—may cause injuries to the cheeks, chin, or bridge of the nose.
Prevention: Placing silicone border or hydrocolloid dressings over bony prominences or other areas that come in contact with the device, and between the device and the skin, has been proven to decrease MDRPIs.
Comment: According to a June 2015 presentation from the Collaborative Alliance for Nursing Outcomes, Memorial Health System in California found that implementing PI prevention and using siliconebased dressings reduced the incidence of stage 3+ MDRPIs per 1,000 patient days from 0.06% to zero in children (benchmark 0.0%- 0.04%) and from 0.28% to zero in adults (benchmark 0.05%-0.09%). The benchmarks used for the study are a comparison to other similar acute care facilities with 500+ beds.
Casts and braces
Casts and braces, as well as cervical collars and stabilizers, put patients at risk for PIs directly under the rim of the device.
Prevention: A study by Browning and Williams found that the following steps help prevent MDRPIs related to casts, braces, cervical collars, and stabilizers.
- Choose the correct device size and position it properly.
- Cushion and protect skin with dressings or silicone pads in high-risk areas.
- Use vigilance around hard-to-see areas (scalp or hairline, under dressings).
- Avoid placing devices over sites of prior or existing injury.
- Be aware of current edema and developing edema under the device.
- Educate staff about correct device use and skin breakdown prevention.
- Remove or reposition devices and provide basic skin care under them.
- Regularly reposition the device, if possible, and record on care and comfort chart.
- If possible, ensure that there’s no contact between the patient’s body and the device.
- Instruct patients and families to inform staff of any discomfort.
Comment: Another measure implemented in the Browning and Williams study included wrapping a red band around the top and bottom of a patient’s plaster cast to alert staff that he or she is at risk of developing a PI and needs specific care.
Tracheostomy flange/ties or securement devices
The patient’s soft tissue under the tracheostomy sheath is at risk for PI.
Prevention: A study by O’Toole and colleagues found that standardizing postoperative procedures reduces the incidence of tracheostomy-related PIs. The protocol includes:
placing a hydrocolloid dressing under the tracheostomy flange during the postoperative period
- removing plate sutures within 7 days of the procedure
- placing a polyurethane foam dressing after suture removal
- placing the patient’s head in a neutral position.
Comment: In the O’Toole study, the incidence of tracheostomy-related MDRPI decreased from 20 of 183 tracheostomies (10.93%) to two of 155 (1.29%). After implementing this protocol at my organization (Michael E. DeBakey VA Medical Center), we’ve achieved zero MDRPI from April 2017 to date.
Catheters
Catheters (urinary, arterial, venous), nasogastric/oral gastric tubes, and fecal management systems present PI risk for any soft tissue that comes into direct contact with these devices.
Prevention: Preventing PIs related to catheters requires:
- assessing for risk factors (sensory deficit, edema, inadequate equipment selection, device positioning or securement, and impaired perfusion)
- using an approved securement device to avoid pressure, trauma, or dislodgment and following the manufacturer’s instruction for indications, monitoring, application, and removal
- inspecting skin under and around the device at least daily
- positioning tubing so that it’s not under the patient or pannus
- educating staff on correct device use and skin breakdown prevention
- checking site frequently for moisture.
Comment: Identifying the possible affected area when perform – ing the assessment is imperative; the assessment should include the skin’s condition and whether there are areas that will be more susceptible to pressure from the catheter.
Sequential compression devices
Improper placement of sequential compression devices (SCDs) can cause injury to the lower extremities.
Prevention: To prevent injuries from SCDs, perform a baseline assessment that includes:
- presence and level of pain
- pallor
- palpable or Doppler pulses
- paresthesia (“pins and needles” sensation)
- paralysis (weakness or lack of movement)
- skin abnormalities under the sleeve
- pain associated with movement or touch
- increasing extremity edema
- signs or symptoms of possible blood clots to the extremity (swelling, redness, pain)
- circumference of the extremity (one size does not fit all).
Comment: Perform routine skin assessments per the manufacturer’s recommendations, and follow placement instructions to ensure that the SCD doesn’t kink, fold, or cause areas of high pressure (check for these issues each time the SCD is activated). Remove SCD sleeves every 8 hours and when bathing, checking for erythema and excessive edema. To protect the skin from increased moisture, use an approved moisture barrier.
What happens if…
Despite best efforts, sometimes a PI may occur. Stage MDRPIs using the standard PI staging system. Exceptions to this rule are mucosal PIs (tongue, GI tract, inner nasal passages, urinary tract, and the vaginal canal), which usually are the result of devices such as oxygen tubing, endotracheal tubes, bite blocks, orogastric and nasogastric tubes, urinary catheters, and fecal containment devices. Al though these injuries may be caused by pressure, there’s no way to differentiate depth, and mucosal injuries typically resolve with no scarring.
It takes a team
It takes a team of devoted staff members, supervisors, nursing executives, and wound care specialists to ensure MDRPI prevention. For example, at the Michael E. De- Bakey VA Medical Center, respiratory technicians play an integral role in prevention by initiating protective measures whenever a CPAP, Bi- PAP, endotracheal tube, or nasal cannula is in use.
Pay attention
All medical devices that might touch patients’ skin, including many not discussed in this article (for example, bed pans and prosthetic limbs), require special attention to address the underlying factors that may lead to an MDRPI. Implementing PI prevention in your practice and organization can reduce injuries and improve patient care.
Selected References
Aronow HN. CALNOC reports: Turning data into information for decision support. October 26, 2015.
Black J, Kalowes P. Medical device-related pressure ulcers. Chronic Wound Care Management and Research. 2016;3:91-99.
Browning R, Williams A. Avoiding pressure ulcers from plaster casts. Nurs Times. 2016;13:7-9.
Edsberg LE, Black JM, Goldberg M, McNichol L, Moore L, Sieggreen M. Revised National Pressure Ulcer Advisory Panel pressure injury staging system. J Wound Ostomy Continence Nurs. 2016;43(6):585-97.
Holden-Mount S, Sieggreen M. Medical device-related pressure ulcers: Pediatrics & adults. 2015.
Joint Commission, The. Quick Safety 43: Managing medical device–related pressure injuries. 2018.
Kayser SA, VanGilder CA, Ayello EA, Lachenbruch C. Prevalence and analysis of medical device-related pressure injuries: Results from the International Pressure Ulcer Prevalence Study. Adv Skin Wound Care. 2018;31(6):276-85.
Lukose M, Thomas L, Camacho-Del Rio G, et al. No more sore ears: Nasal cannula related pressure ulcers down to zero. WOCN 49th Annual Conference. 2017.
National Database of Nursing Quality Indicators® (NDNQI®). Pressure injuries and staging: Medical device related (MDR) pressure injuries.
National Pressure Ulcer Advisory Panel. Best Practices for Prevention of Medical Device-Related Pressure Injuries. Updated May 2017.
National Pressure Ulcer Advisory Panel. Mucosal Pressure Ulcers: An NPUAP Position Statement.
O’Toole TR, Jacobs N, Hondorp B, et al. Prevention of tracheostomy-related hospital-acquired pressure ulcers. Otolaryngol Head Neck Surg. 2017;156(4):642-51.
Wound Ostomy and Continence Nurses Society® (WOCN). (2017). WOCN Society position paper: Avoidable versus unavoidable pressure ulcers (injuries). Mt. Laurel, NJ: WOCN.
Gustavo Camacho-Del Rio is a wound specialist on the skin risk management team at Michael E. De- Bakey VA Medical Center in Houston, Texas.
ant10-Pressure Injuries-918
















1 Comment.
Hi I would like to know which soft silicone Nasal Cannula you are using in your institution. Were there any contraindications or events when you switched to this NC.
Do you always place a Silicone foam or Hydrocolloid to ears, cheeks when using high flow O2, Bipaps?